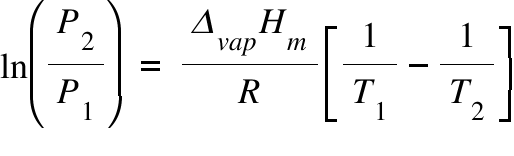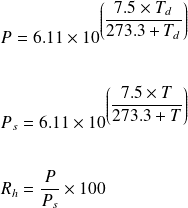The vapour pressure of pure water is 760 mm at 25^°C . The vapour pressure of solution containing 1(m) solution of glucose will be ??
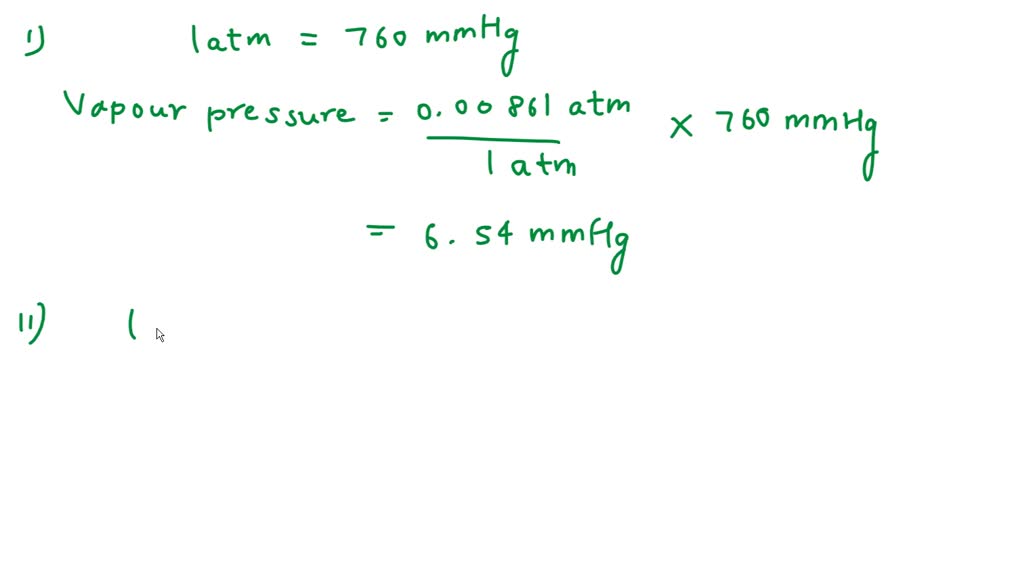
SOLVED: The vapor pressure of water at 5°C is 0.00861 atm. Calculate the vapor pressure in mmHg and torr. Round each of your answers to 3 significant digits. mmHg? torr?

The vapour pressure of a pure liquid 25°C is 100 mm Hg. Calculate the relative lowering vapour pressure the mole fraction of solvent in solution is 0.8. pº - PS = Xsolute