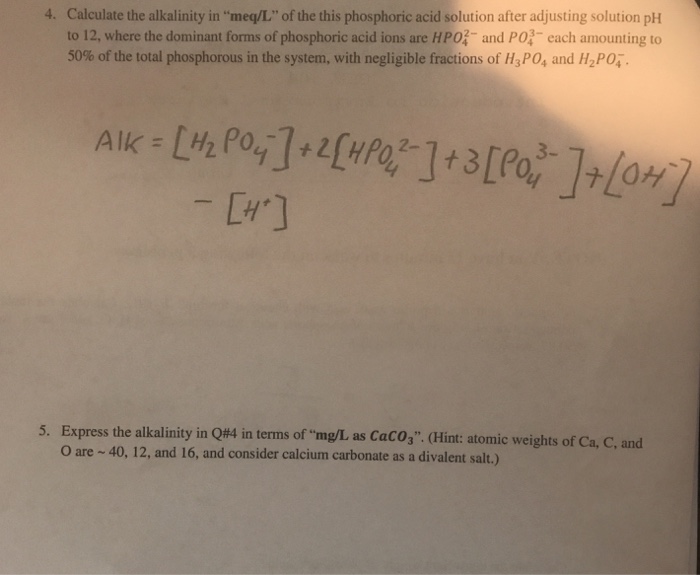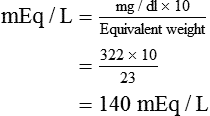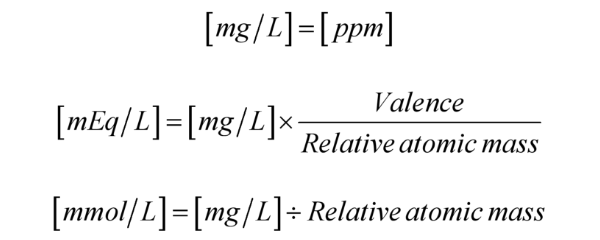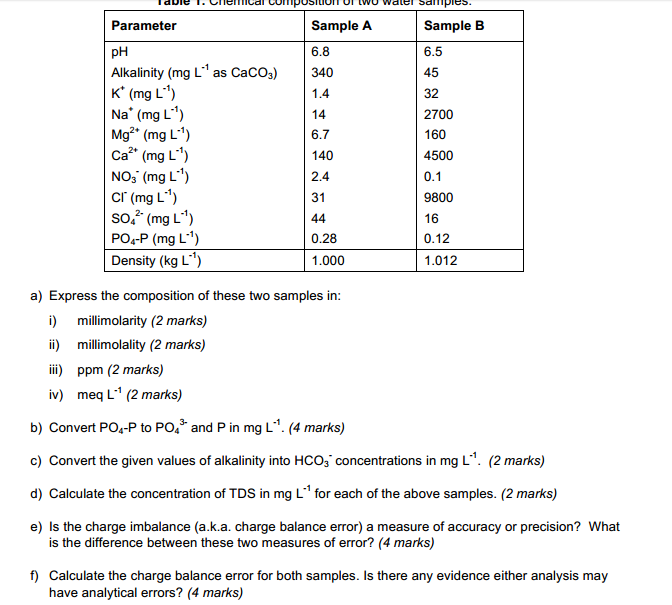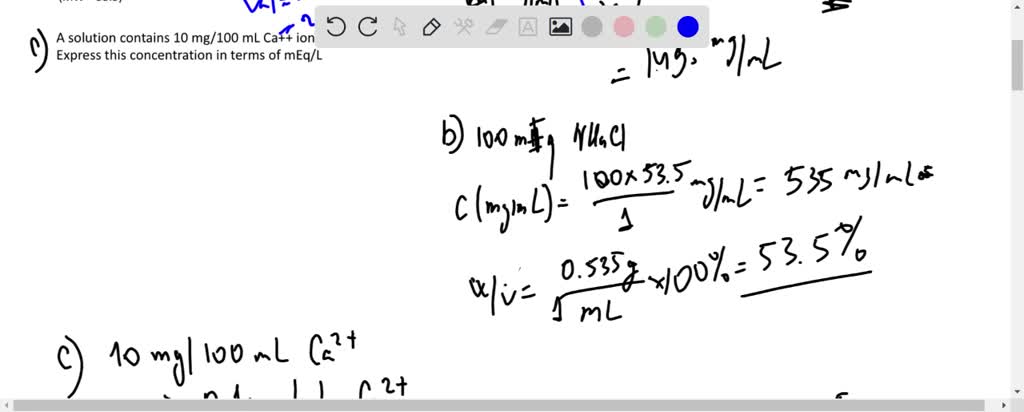
SOLVED: What is the concentration, in mg/mL, of a solution containing 2 mEq of potassium chloride (KCl) per mL? What is the percent (w/v) concentration of a solution containing 100 mEq of
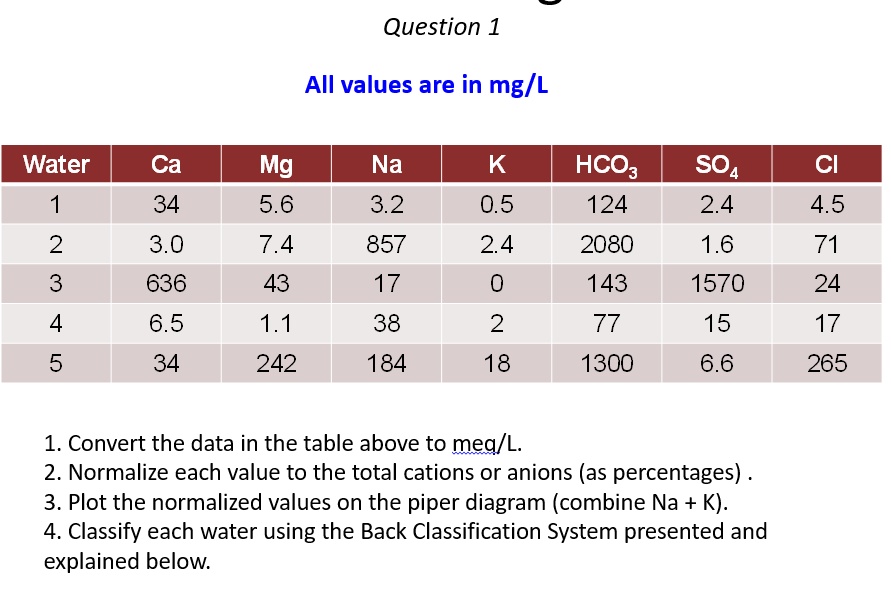
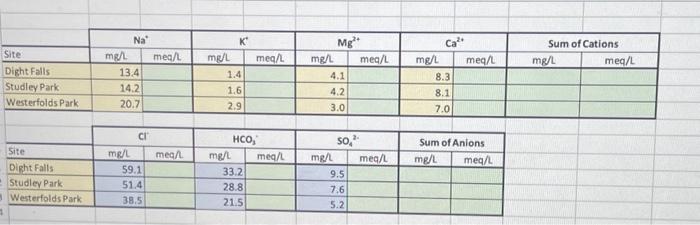
SOLVED: Question 1 All values are in mg/L Water Ca Mg 5.6 7.4 43 1.1 242 Na K Cl 34 3.2 0.5 2.4 0 2 HCO3 124 2080 143 77 1300 SOa